Dynamic Charge Acceptance (DCA) testing of cells
The Dynamic Charge Acceptance (DCA) of a cell is a measurement of the battery / cells ability to absorb electrical charge in relation to the capacity of the battery.
This parameter is particularly important in the case of electric and hybrid electric vehicles, where the ability of the vehicle battery to accept energy determines the amount of energy which can be recovered from the vehicle as it slows down. The DCA can be a function of battery chemistry, battery temperature, state of charge (SoC) and usage history of the battery / cell.
To best match real-world operating conditions whilst maintaining consistency during the test procedure, all testing has been performed under temperature controlled conditions, with the cells being placed in free air, within temperature controlled chambers, this method of testing having the advantage that it has been possible to perform tests below 0 ⁰C.
Testing has been performed at -10, 0, 10, 25 & 40 ⁰C. This range of temperatures covers the normal range of temperatures which may be expected to occur in real-world service within EVs.
An advanced battery and cell testing system has been used to conduct DCA tests on the cells at various SoC and charge history; specifically: 90, 70, 30 & 10 % SoC with the battery / cell having a discharge history, and 10, 30, 50, 70 & 90 % SoC with the battery / cell having a charge history.
Tests have been carried out on various chemistries, including Lithium based cells and advanced Lead based cells to determine their characteristics. The results of the tests are scales to the cell capacity ‘C’ to ensure the ability to compare like with like.
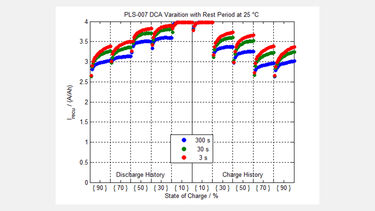
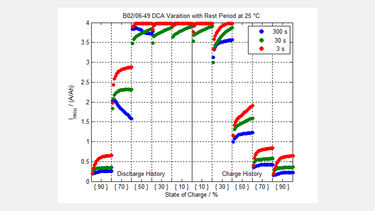
A typical result from an advanced lead based cell (B02/06-49) and a Lithium iron phosphate cell (PLS-007) are shown for comparison in the figures.
These show how the cells behave for each one of 20 pulses of energy pushed back into the cell, at 25oC, and at various cell states of charge, and both previous charge and discharge history for the cell.
The maximum value of the energy was limited by limiting the current to 4 x the ‘C’ rate of the cell in each case. It is interesting to note from the results, that it may not be clear cut as to the choice between Lithium based batteries and Lead based batteries for some applications.